

Proteomics Experiment #2
Application of in–source fragmentation (cone voltage fragmentation) for structure elucidation by the Finnigan Navigator MS Spectrometry
Wendy W.Y. Zhang, Sazzad Hussain, Qianfan Wang, Analytical Science Department, Shionogi BioResearch Corp., 45 Hartwell Ave. Lexington, MA 02421
Introduction
MS spectrometry with atmospheric pressure ionization (API) is a one of the most powerful analytical tools in pharmaceutical industry. The API is a soft ionization technique providing molecular weight information. However, if increasing the voltage on the sample cone, fragmentation of parent ion could occur, which is also called in-source fragmentation. By utilizing the in-source fragmentation technique structural information can be obtained. In this poster, we have reported three cases regarding fragmentation by the in-source fragmentation technique.
Materials and Methods
Three compounds, TP89128, 11-3423E, and 11-3428E, were made in house. Analysis of these samples was performed on a Finnigan Navigator LC/MS system. An APcI probe with positive ion mode, full scan, probe temperature 550oC, and source temperature 120oC was applied.
Results
At first, sample TP89128 (exact mass 501.2) was analyzed at cone voltage 5 and 25 respectively. In this case we observed a relatively high intensity molecular ion peak at m/z 502.3 with two of its fragments at m/z 446.2 and 402.2 at cone voltage 5. When the cone voltage was increased to 25, the relative intensity of molecular ion peak drastically decreased and additional fragment at m/z 366.3 was showed up due to elimination of Cl from the fragment of [M - tBu- COOH + H+]. The table 1 shows the possible fragments.
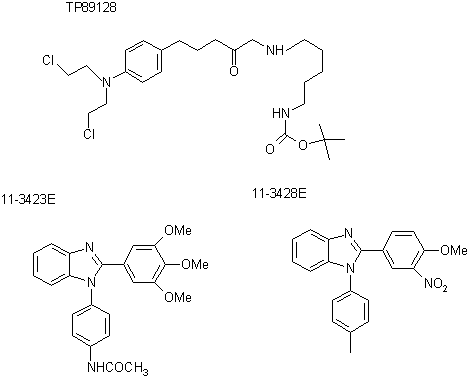
Table 1: Fragmentation of TP89128
| Cone Voltage 5 | Cone Voltage 25 | ||
| Ion | M/z | Ion | M/z |
| M + Na+ | 524.3 | M + Na+ | 524.2 |
| M + H+ | 502.3 | M + H+ | 502.3 |
| M – tBu + H+ | 446.2 | M – tBu + H+ | 446.2 |
| M – tBu - H2O + H+ | 428.1 | M – tBu - H2O + H+ | 428.2 |
| M – tBu –COOH + H+ | 402.2 | M – tBu – COOH + H+ | 402.2 |
| M – tBu – COOH – Cl + H+ | 366.3 | ||
Second, sample 11-3423E (exact mass 417.2) was analyzed using cone voltage, 10, 20, 40, 60 and 80 respectively. In this case we did not see any significant fragmentation at cone voltage 10, 20, and 40. At cone voltage 60 a fair amount of fragmentation was observed, which increased when cone voltage elevated to 80. Based on test results, possible fragments are listed in the table 2.
Table 2: Fragmentation of 11-3423E
| Cone Voltage 10 | Cone Voltage 60 | ||
| Ion | M/z | Ion | M/z |
| M + Na+ | 440.2 | M + Na+ | 440.2 |
| M + H+ | 418.3 | M + H+ | 418.3 |
| M - COCH2 + H+ | 376.2 | M - COCH2 + H+ | 376.2 |
| M - COCH2 - OCH2 + H+ | 436.1 | ||
| M - COCH2 - 3OCH2 + H+ | 286.1 | ||
Then 11-3428E (exact mass 417.3) was also analyzed using cone voltages, 10, 20, 40, 60 & 80 respectively. No any significant fragments at cone voltage 10 and 20 were observed, but we observed a large number of fragments at cone voltage 40, 60, and 80 respectively. The most fragments were observed at cone voltage 80. The fragments are shown in the table 3.
Table 3: Fragmentation of 11-3428E
| Cone Voltage 10 | Cone Voltage 60 | ||
| Ion | M/z | Ion | M/z |
| M + Na+, M + H+ | 428.2, 406.2 | M + Na+, M + H+ | 428.2 |
| M - NO2 + H+ | 359.1 | ||
| M - NO2 - CH2 + H+ | 343.2 | ||
| M - NO2 - OCH2 + H+ | 329.1 | ||
Conclusion:
The in-source fragmentation is a useful technique, which can provide the information of structures of compounds. If increasing the voltage on the sample cone, fragmentation of parent ion could occur. With this information of the fragmentation, we will be able to elucidate and confirm the structures of compounds